|
UV-vis
spectroscopy is usually used for molecules, inorganic
ions or complexes in solution. A UV-vis
Spectrophotometer exposes a sample to the ultraviolet region
of the electromagnetic spectrum. Depending on the sample
being used, a certain amount of the light is absorbed which
results in the excitement of electrons. A spectrum is then
made from the light that reaches the detector.
UV spectroscopy is
based on Beer’s Law.
The Beer-Lambert Law states that
A = ebc,
Where A is absorbance (no units),
b is the path length (cm) which is usually 1cm, c is the concentration (mol L-1)
and e is a constant of
proportionality (L mol-1
cm-1)
called the absorbtivity.
this equation can also be written as
A = e
l c
where e is
the molar absorptivity which is sometimes called the extinction
coefficient (variation of e
will occur with wavelength of
light used). l is the path length and c is the concentration.
These two equations
tell us that absorbance depends on
the total quantity of the absorbing compound in the light path
through the cuvette. (http://www.shu.ac.uk/schools/sci/chem/tutorials/molspec/beers1.htm)
By measuring A
and knowing I and
e
the concentration, C
can be calculated.
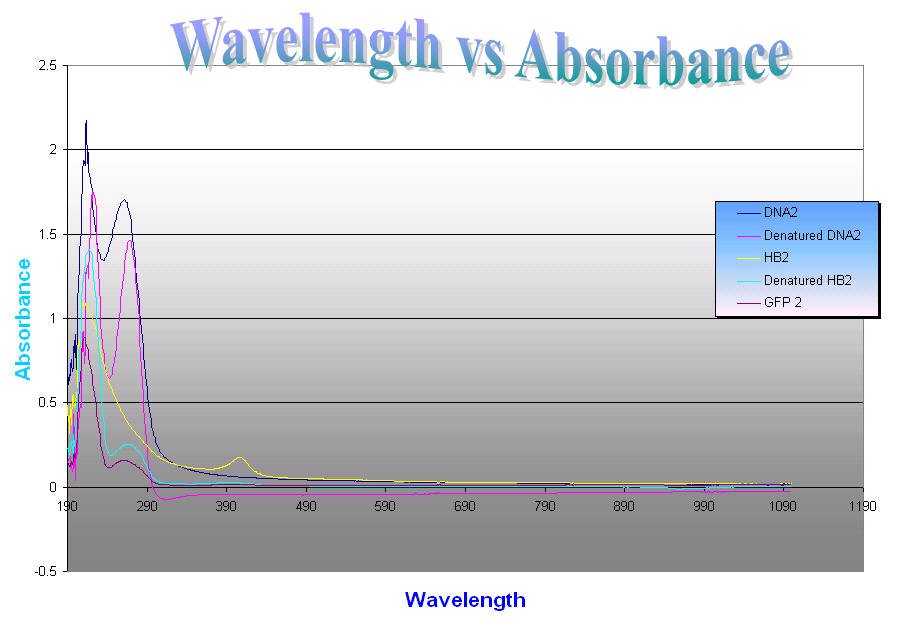
Different wavelengths are absorbed by
different molecules. Proteins absorb ultraviolet light at the range of 190-300
nm although proteins can be detected in the range from 190-240, this wavelength
range is called the far UV. The amide bond is found in this region with a
maximum absorption at 214 nm. Proteins also absorb light in the near UV which
is the range from 250-300 nm. At this range absorptions of the aromatic
amino acids tryptophan, tyrosine, and phenylalanine and disulfide bonds occur.
Although when measuring protein concentration, the absorption at 280nm is
usually used. And DNA has a maximal
absorption peak at 260nm . |